Curious about why carbon can form millions of compounds while most of the other elements barely make a few? The secret lies in organic chemistry bonding the invisible framework that builds every molecule around you, from the DNA in our cells to the caffeine in our morning coffee.
Understanding atoms is not just about memorizing formulas. It’s about seeing electron clouds, feeling how orbitals overlap, and visualizing why molecules twist, bend, and react. Hybridization and molecular geometry are not just abstract ideas; they explain why diamonds are hard, gasoline burns, and our phone screen responds to our touch.
In this deep dive, we will explore sigma and pi bonds, decode hybridization from sp³ to sp, and uncover how electron density maps reveal the real nature of chemical bonding. Whether you are aiming to ace organic chemistry or are simply curious, this guide will change the way you see molecules forever.
Covalent Bonds: A Quick Refresher
Covalent bonds happen when atoms share electrons to become stable. But here’s the twist textbooks often miss: electrons don’t just sit between atoms like tiny planets. The real magic is orbital overlap, when atomic orbitals merge to form a shared space for electrons.
Imagine two soap bubbles touching. The area where they overlap? That’s your bond. The bigger the overlap, the stronger the connection. This shared space packs electron density right between the nuclei, holding them together with electrostatic attraction.
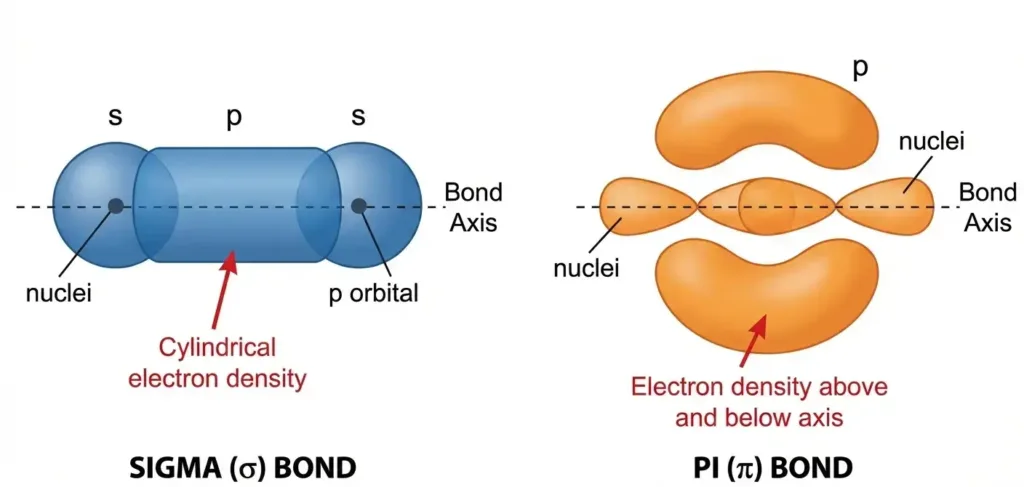
Sigma (σ) vs. Pi (π) Bonds
There are two fundamental types of covalent bonds, and understanding their difference is crucial for mastering organic chemistry bonding:
Sigma (σ) bonds form through head-on orbital overlap. Picture two orbitals meeting face-to-face along the axis connecting the atoms. This creates a cylindrical cloud of electron density directly between the nuclei. Every single bond you see is a sigma bond.
Pi (π) bonds form through side-by-side overlap of p orbitals. Imagine two parallel orbitals touching along their sides, creating electron density above and below the bonding axis. Pi bonds always come as additions to sigma bonds in double and triple bonds.
Here’s the visual breakdown:
- Single bond = 1 σ bond
- Double bond = 1 σ bond + 1 π bond
- Triple bond = 1 σ bond + 2 π bonds
The sigma framework acts as the skeleton of organic molecules, while pi clouds add reactivity and rigidity.
Why Sigma Bonds Allow Rotation, Pi Don’t
This is where it gets fascinating. Sigma bonds allow free rotation because their electron cloud is evenly spread around the bond axis. Twist a single bond all the way around, and the orbital overlap stays the same; nothing snaps.
Pi bonds, on the other hand, are stuck. Their side-by-side p orbital overlap would break if the bond rotated, destroying the connection. That’s why double bonds make molecules rigid and give rise to cis-trans isomers. Think of the pi cloud like a molecular handcuff, locking the shape in place. Recognizing how bonds rotate is key to predicting how molecules twist, fold, and react in organic chemistry.
Hybridization Theory: From Orbitals to Shapes
In the 1930s, Linus Pauling cracked a big mystery: if carbon has two types of atomic orbitals—one s and three p—why do its four bonds in methane all look the same?
His answer was hybridization. Before bonding, atomic orbitals combine mathematically to form new, equivalent hybrid orbitals. This isn’t a physical mix; it’s a quantum idea that predicts molecular shapes and bonding patterns more accurately.
Valence bond theory uses hybridization to explain molecule shapes, while molecular orbital theory shows the full electronic structure. Both are correct, but hybridization is easier to grasp, especially when studying organic chemistry bonding.
sp, sp², sp³ Explained with Shapes & Angles
To understand hybridization explained for the organic world, we look at how many “electron groups” (atoms or lone pairs) surround an atom.
sp³ Hybridization: One s orbital + three p orbitals = four equivalent sp³ orbitals arranged tetrahedrally. Bond angle: 109.5°. This is your standard carbon in alkanes, with maximum electron repulsion balance according to VSEPR theory.
sp² Hybridization: One s orbital + two p orbitals = three sp² orbitals in a trigonal planar arrangement. Bond angle: 120°. One p orbital remains unhybridized and perpendicular to the plane, ready to form pi bonds. This is the carbon in alkenes.
sp Hybridization: One s orbital + one p orbital = two sp orbitals in a linear arrangement. Bond angle: 180°. Two p orbitals remain unhybridized for forming two pi bonds. This is the carbon in alkynes.
The pattern? More s character means orbitals hold electrons closer to the nucleus, making atoms more electronegative and bonds stronger and shorter.
Hybridization Types in Organic Chemistry
| Hybridization | Orbitals Mixed | Number of Hybrid Orbitals | Molecular Shape | Bond Angle | Unhybridized p Orbitals | Typical Organic Example |
|---|---|---|---|---|---|---|
| sp³ | 1 s + 3 p | 4 sp³ | Tetrahedral | 109.5° | 0 | Alkanes (e.g., methane, ethane) |
| sp² | 1 s + 2 p | 3 sp² | Trigonal planar | 120° | 1 | Alkenes (e.g., ethene) |
| sp | 1 s + 1 p | 2 sp | Linear | 180° | 2 | Alkynes (e.g., ethyne) |
Lone Pairs and Deviations
Hybridization isn’t just about bonding orbitals—lone pairs play a role too. They occupy hybrid orbitals and influence molecular shapes by repelling other electrons.
Take ammonia (NH₃) as an example. Its nitrogen is sp³ hybridized with one lone pair. Normally, a tetrahedral angle would be 109.5°, but the lone pair pushes harder than bonding pairs. This compresses the H–N–H angle to 107°.
Water (H₂O) takes it a step further. Oxygen is sp³ hybridized with two lone pairs. These lone pairs push strongly, squeezing the H–O–H angle to 104.5°. The molecule forms a bent shape, not a perfect tetrahedron, because lone pairs dominate the repulsion forces.
This shows that hybridization predicts how orbitals arrange themselves. VSEPR theory fine-tunes the molecular geometry. It does so by accounting for electron-electron repulsion.
Electron Clouds & Bonding Patterns in Organic Chemistry Bonding
We often draw electrons as dots, but in reality, they exist as a “cloud” or electron density.
1. Electron Density Maps
Electron density maps show where electrons actually hang out. Instead of rigid sticks, bonds appear as fuzzy clouds with areas of high and low concentration.
When electron density is high between nuclei, the bond is strong. Low density points to weak interactions or empty regions. Chemists now use these maps to predict reactivity, acidity, and likely reaction sites.
Looking at a molecule’s electron cloud is like seeing the real story behind Lewis structures. Electrons behave like waves, not tiny particles, and their arrangement controls everything—from stability to color.
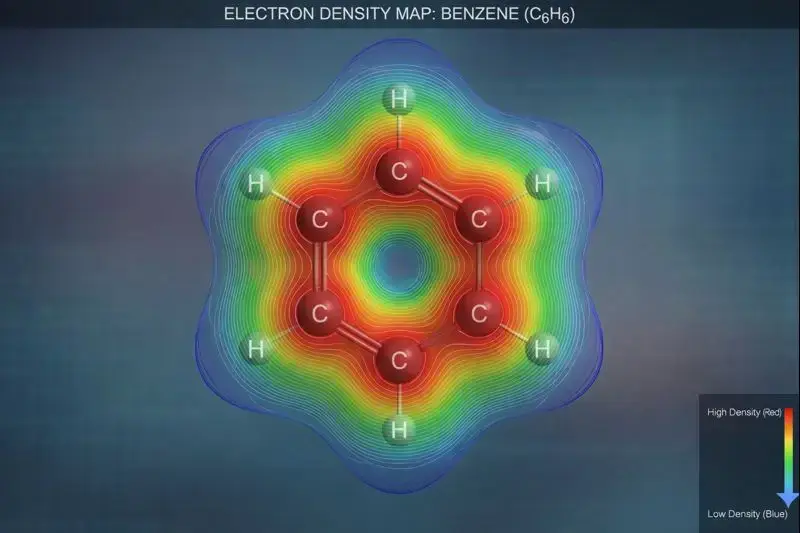
2. How Electron Clouds Affect Stability & Geometry
Electron clouds seek the lowest energy configuration. This drives molecular geometry through:
Resonance stabilization: Delocalized pi clouds spread electron density across multiple atoms, lowering energy. Benzene’s six-electron aromatic system is more stable than three separate double bonds.
Electronegativity effects: Electron density shifts toward more electronegative atoms, creating polar bonds and dipole moments that influence shape and reactivity.
Steric repulsion: Electron clouds physically push against each other, determining bond angles and molecular conformations.
The overall geometry emerges from balancing orbital overlap (maximizing bonding) with electron repulsion (minimizing repulsion).
Organic Examples & Case Studies
Let’s apply these concepts to the celebrities of the organic world.
1. Alkanes (sp³): The Tetrahedral Standard
Methane (CH₄) is the perfect sp³ system. Four identical C-H sigma bonds point toward the corners of a tetrahedron. Each bond has 25% s character and 75% p character, creating bonds of equal strength and length (1.09 Å).
Ethane (C₂H₆) adds complexity with a C-C sigma bond that allows rotation. This creates infinite conformations—eclipsed (higher energy) and staggered (lower energy)—all accessible through rotation around the sigma framework.
The tetrahedral geometry of sp³ carbon is why alkanes form three-dimensional structures and why life’s molecules have such diverse shapes.
2. Alkenes (sp²): The Planar Powerhouse
Ethene (C₂H₄) introduces sp² hybridization. Each carbon has three sp² orbitals forming sigma bonds (one C-C, two C-H) in a flat, 120° arrangement. The unhybridized p orbitals overlap side-by-side, creating the pi bond above and below the plane.
This pi cloud locks the molecule flat. You can’t rotate around the C=C double bond without breaking the pi overlap. The result? Geometric isomers (cis/trans) and a reactive site where electrophiles attack the electron-rich pi system.
The planar geometry and restricted rotation of alkenes drive much of organic reaction chemistry, from addition reactions to polymer formation.
3. Alkynes (sp): The Linear Link
Acetylene (C₂H₂) takes linearity to the extreme with sp hybridization. Each carbon has two sp orbitals forming sigma bonds (one C-C, one C-H) at 180°. Two perpendicular p orbitals on each carbon create two pi bonds, forming a cylindrical electron cloud around the C≡C axis.
The 50% s character in sp orbitals makes acetylene’s C-H bonds more acidic than alkanes or alkenes. The electrons are held tighter, making the hydrogen more easily removed as H⁺.
This linear geometry and high electron density between carbons make alkynes versatile building blocks in organic synthesis.
Benzene & Aromaticity (Delocalized π Clouds)
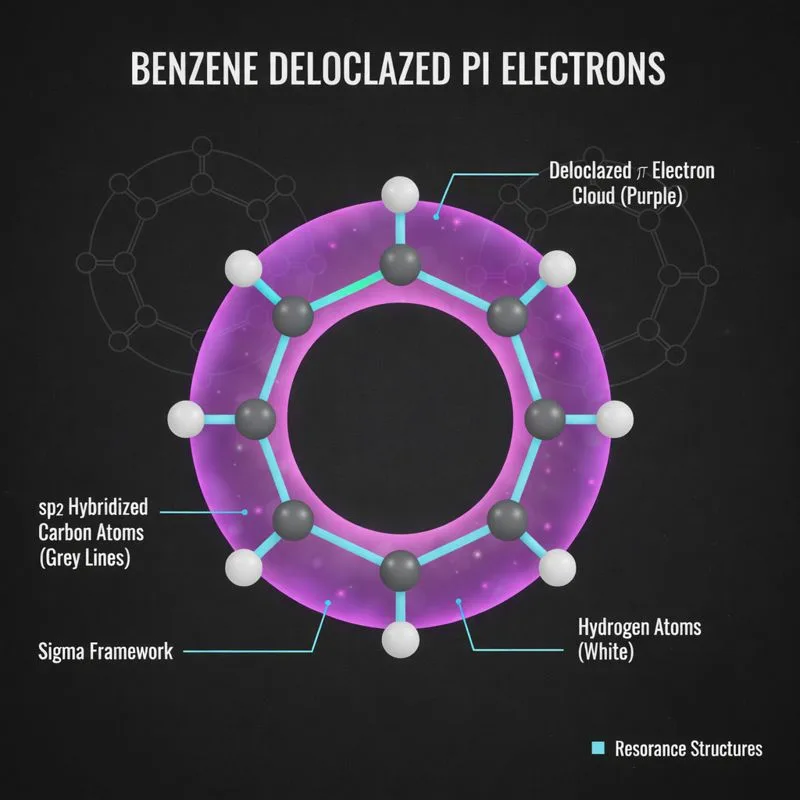
Benzene (C₆H₆) revolutionizes bonding with its delocalized pi system. Six sp² carbons form a perfect hexagonal ring with sigma bonds. Six p orbitals contribute six pi electrons that don’t belong to any single bond; they spread evenly across the entire ring.
This delocalization creates exceptional stability called aromaticity. The electron cloud sits above and below the ring plane like two donuts, with uniform electron density. This explains why benzene doesn’t behave like three separate double bonds. It also resists addition reactions that would disrupt aromaticity.
Aromatic stabilization is worth about 36 kcal/mol, a huge energetic advantage that makes benzene rings ubiquitous in nature and chemistry.
Hybridization and Reactivity in Organic Chemistry Bonding
Why does hybridization matter for your exams? Because it predicts reactivity.
Nucleophiles, Electrophiles & Orbital Character
Chemical reactivity depends on the type of orbital an atom has. Nucleophiles carry high-energy electron pairs and look for electron-poor spots. Electrophiles, on the other hand, have empty, low-energy orbitals ready to accept electrons.
sp³ carbons have low s-character, making them weaker nucleophiles but giving stable leaving groups. sp² carbons form reactive pi bonds that attract electrophiles easily. sp carbons, with high s-character, hold electrons tightly, which changes both their nucleophilicity and acidity.
By understanding hybridization, you can predict where and how reactions happen. Pi bonds often act as nucleophiles during addition reactions. Lone pairs in high-energy sp³ orbitals are more reactive than those in lower-energy sp² orbitals.
Why sp Hybrid Carbons Are More Acidic
Here’s a game-changer: terminal alkynes (R-C≡C-H) are about 10²⁵ times more acidic than alkanes. Wild, right?
The secret lies in the sp hybrid orbital. With 50% s character, its electrons hug the nucleus much closer than in sp³ (25% s) or sp² (33% s). When the hydrogen leaves as H⁺, the negative charge that’s left behind sits in the sp orbital. This makes it more stable—lower energy, tightly held, and less eager to grab the proton back.
This link between electronegativity and hybridization drives so much of organic chemistry. It explains everything from acid-base reactions to organometallic synthesis, helping us predict how molecules behave.
Special Organic Chemistry Bonding Cases You Need to Know
Sometimes, molecules don’t follow the “standard” rules.
Bent Bonds in Small Rings
Not all molecules follow textbook geometry. Small rings, like cyclopropane, feel extra stress because their bond angles can’t reach the ideal values. In cyclopropane, sp³ carbons are forced into 60° angles instead of the usual 109.5°. This creates “bent bonds,” where orbitals overlap less efficiently, raising the molecule’s energy.
To cope, the ring increases p character in its bonds while the outer C-H bonds gain more s character. This built-in strain makes small rings highly reactive. That’s why three- and four-membered rings easily undergo ring-opening reactions, releasing the stored energy.
Hyperconjugation & Resonance Effects
Hyperconjugation occurs when sigma bond electrons interact with adjacent empty or partially filled orbitals. A C-H sigma bond can donate electron density into a neighboring empty p orbital or pi system, stabilizing positive charges or radicals.
Resonance stabilization delocalizes electrons across multiple atoms through pi systems. Carboxylate ions spread negative charge across two oxygens. Allylic carbocations distribute positive charge across three carbons.
Both effects demonstrate that electrons don’t stay localized; they find lower energy states through orbital interactions that Lewis structures alone can’t show.
Spectroscopy and Bond Types in Organic Chemistry Bonding
We can actually see these bonds using technology.
How σ/π Bonds Appear in IR/NMR/UV-Vis
Different bonds absorb different wavelengths of electromagnetic radiation, making spectroscopy the ultimate bonding detective:
Infrared (IR) Spectroscopy: Sigma bonds between different atoms create characteristic stretching frequencies. C-H stretches appear around 2900-3000 cm⁻¹. C=O double bonds (σ + π) show strong absorptions near 1700 cm⁻¹. Triple bonds push even higher.
NMR Spectroscopy: Chemical shifts reveal electron density. Electrons in pi clouds create local magnetic fields that shield nearby nuclei differently than sigma electrons, shifting resonance frequencies.
UV-Vis Spectroscopy: Pi electrons transition from bonding to antibonding orbitals when absorbing UV or visible light. Conjugated systems with multiple pi bonds show progressively red-shifted absorptions.
Practical Examples
Analyzing an unknown compound? IR shows a peak at 2100 cm⁻¹—likely a C≡C or C≡N triple bond. NMR shows a peak far downfield at 7-8 ppm—probably aromatic hydrogens in a benzene ring. UV-Vis shows absorption at 450 nm—an extended conjugated system with multiple pi bonds.
Each spectroscopic technique reveals different aspects of orbital structure, and together they provide a complete picture of organic chemistry bonding.
Molecular Orbital Perspective Beyond Valence Bond Theory
While valence bond theory is great for shapes, molecular orbital theory is the “pro level.”
Instead of individual atoms, MO theory treats the whole molecule as one big orbital system.
HOMO, LUMO, Bonding/Anti-bonding
Molecular orbital theory takes a different approach than hybridization. Instead of combining atomic orbitals locally, MO theory combines all atomic orbitals across the entire molecule, creating bonding orbitals (lower energy) and antibonding orbitals (higher energy).
The HOMO (highest occupied molecular orbital) represents the most reactive electrons—these initiate most chemical reactions. The LUMO (lowest unoccupied molecular orbital) accepts electrons during reactions.
The HOMO-LUMO gap determines molecular properties like color, reactivity, and conductivity. Small gaps mean lower energy transitions and more reactive molecules.
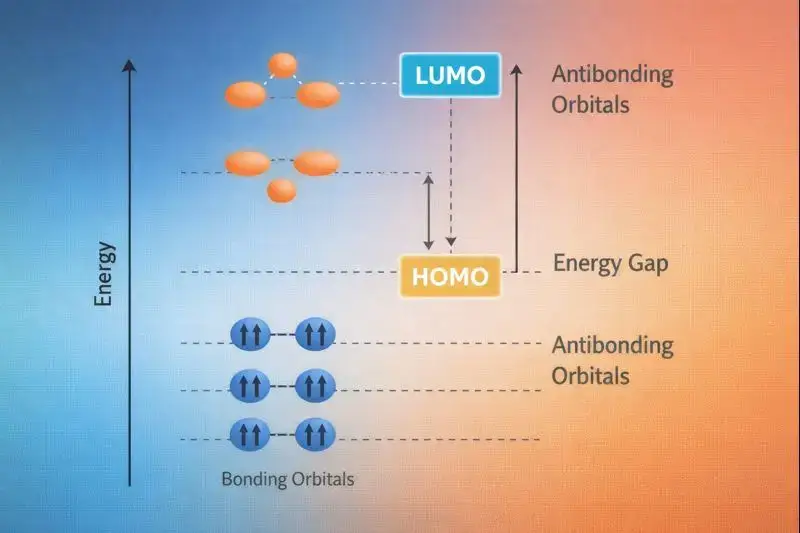
What MO Theory Adds to Hybridization
While valence bond theory with hybridization explains local bonding and geometry, molecular orbital theory reveals:
- Delocalization: Pi systems in benzene are better described by molecular orbitals spanning the entire ring
- Magnetism: Explains why O₂ is paramagnetic despite appearing to have all paired electrons
- Reaction mechanisms: HOMO-LUMO interactions predict reaction pathways and selectivity
- Electronic structure: Shows how electrons occupy energy levels across entire molecules
Both theories complement each other. Hybridization gives intuitive local geometry. MO theory provides global electronic structure.
Conclusion
Understanding organic chemistry bonding turns molecules from abstract formulas into living, three-dimensional structures. Hybridization shows why carbon can build such a variety of forms—from the tetrahedral shapes in life’s molecules to the flat rigidity of double bonds and the straight lines of triple bonds.
Electron clouds are more than just theory. They are invisible forces that decide if a molecule is stable or reactive, twisted or flat, colored or clear. The dance between sigma frameworks and pi systems shapes all of organic chemistry—from the simplest alkane to the most intricate protein.
Take up these ideas—orbital overlap, hybridization, sigma vs. pi bonds, and electron density—and you don’t just learn organic chemistry. You see the molecular world in a whole new way. The hidden life of bonding finally comes into view.
Recommended Resources for Curious Minds
1. Organic Chemistry As a Second Language: First Semester Topics by David Klein
This workbook breaks down complex bonding concepts into digestible practice problems. It’s perfect for reinforcing hybridization. It also enhances understanding of molecular geometry through hands-on exercises.
2. Molecular Models Set (Organic Chemistry Kit)
Building physical models of sp, sp², and sp³ molecules transforms abstract hybridization concepts into concrete realities. This helps you visualize bond angles and molecular geometry in three dimensions.
3. Organic Chemistry, 9th Edition by John McMurry
This textbook is comprehensive. It has exceptional visuals of electron density maps and orbital diagrams. It is ideal for deep dives into bonding theory and spectroscopy.
4. The Organic Chem Lab Survival Manual by James Zubrick
This is a practical lab guide. It connects bonding theory to real-world applications. It shows how sigma and pi bonds affect reactivity in actual experiments.