Approximately millions of cells in our bodies experience a programmed cell death every second. This is the last type of cell destruction. It is somewhat alarming. But don’t worry, it is actually a good thing. This process is essential for keeping us healthy. So, how do these little cells know when it’s their time to die? It is not a pathological process. Instead, it is part of tissue homeostasis. This occurs in a controlled process known as apoptosis. Unlike loss of tissues after trauma or pathological conditions, apoptosis is carried out systematically. It breaks the cell down in both normal and developing cells. It also occurs in cells that are in the turnover process.
Understanding programmed cell death is very important. It gives us clarity into diseases like cancer, brain disorders, autoimmune conditions, and even aging. If cells don’t die when they are supposed to, that is how tumors can form. On the other side, if they kick the bucket too easily, our tissues might not stay strong.
In this article, we will dive into how cells “decide” to meet their end. We will explore the signals that start this process. We will also look at what can go wrong along the way. By the time we wrap things up, you will see that programmed cell death is not a scary thing. It is actually a vital part of what keeps us alive.
Apoptosis 101: Why Cells Need to Die?
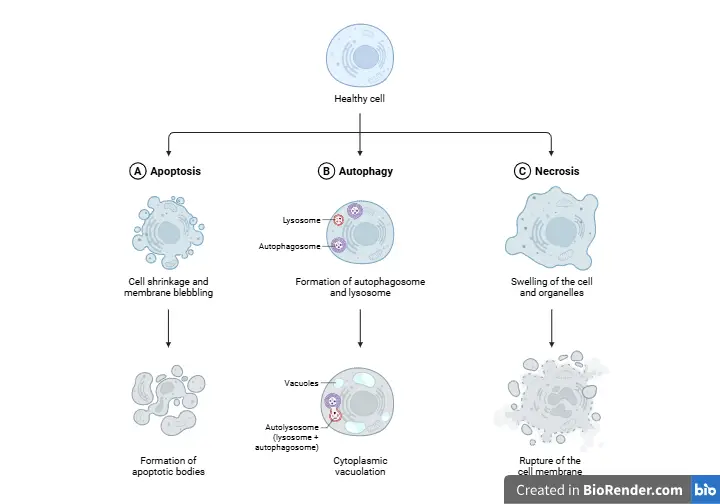
Apoptosis is basically a kind of programmed cell death that happens in a well-planned order. It is completely different from necrosis, which is that chaotic, uncontrolled kind of death that usually happens due to injury. Cells use apoptosis to shape tissues, tackle infections, and clear out damaged components without causing any inflammation. It is like a well-rehearsed performance, keeping everything in check and running smoothly. Now, lets diving into the mechanics.
1. The Biological Imperative: Roles of Apoptosis
Apoptosis, also known as programmed cell death, is an energy-dependent process. It is characterized by genetically programmed cell death. Its indispensableness is because it restores tissue homeostasis by selective cell destruction.
- Development: Apoptosis shapes organs. Tadpoles lose their tails. Human fingers separate as webbing cells self-destruct.
- Tissue Homeostasis: Old blood cells, skin cells, and gut lining cells die on cue. New cells replace them to keep tissues healthy.
- Immune System: Apoptosis removes faulty immune cells and infected cells. This prevents autoimmunity and controls viral spread.
- Disease Prevention: Precancerous cells are eliminated before they form tumors. Cells with severe DNA damage trigger self-destruction.
2. Accidental vs. Programmed: A Crucial Distinction
Apoptosis is often confused with necrosis, but they differ significantly:
| Feature | Apoptosis | Necrosis |
|---|---|---|
| Morphology | Cell shrinkage, chromatin condensation, apoptotic bodies | Cell swelling, membrane rupture, organelle breakdown |
| Energy | ATP-dependent | ATP-independent |
| Inflammation | No | Yes |
| DNA | Fragmented into nucleosome-sized pieces | Random degradation |
| Phagocytosis | Yes, by neighboring cells | No, leads to leakage of contents |
Understanding the difference between these two processes is crucial. If either one gets out of whack, it can lead to some pretty serious health issues.
How Apoptosis is Triggered and Executed
Now we reach the heart of our question: how do cells know when to die? The response is intricately interwoven with molecular signals and pathways. These little guys are like tiny messengers. They can pick up on signs of stress in the cell. They notice changes during development. Furthermore, they can even detect signals from outside the cell. There are two major apoptotic modules that are activated by specific death stimuli.
1. The Extrinsic Pathway: Signals from Without
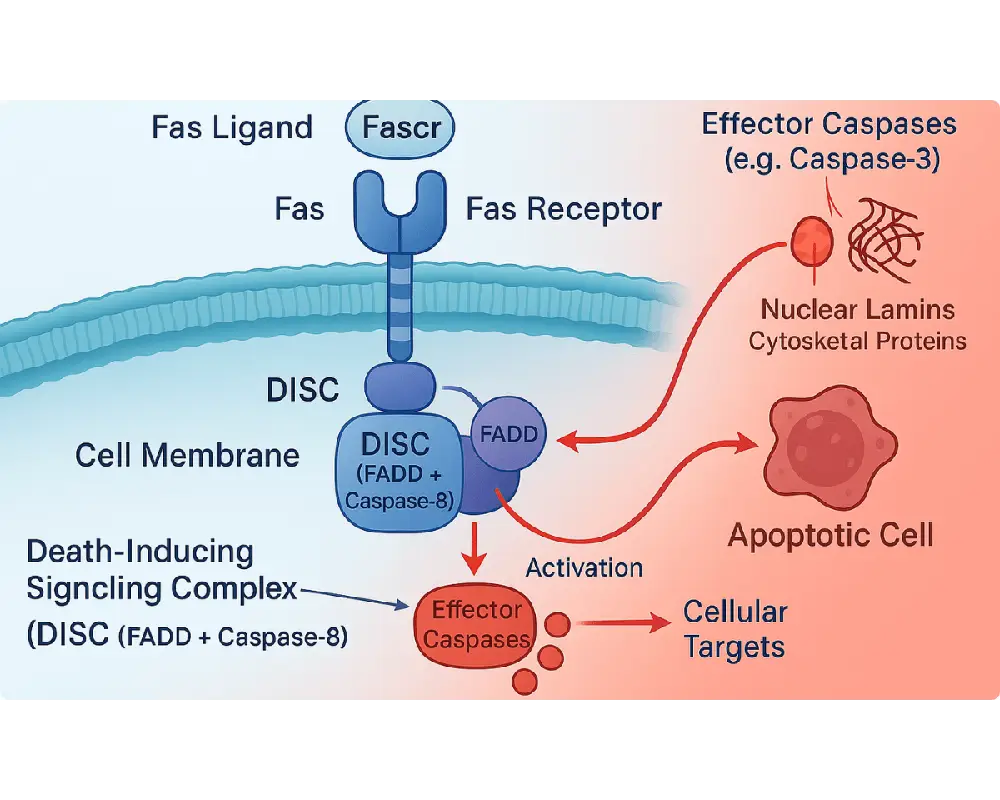
The extrinsic pathway is like a cellular “kill switch” activated by external signals, often from the immune system. It involves:
- Death Receptors: Proteins like Fas (CD95) and TNF receptor 1 (TNFR1) on the cell surface bind ligands (e.g., FasL, TNF-α).
- Death-Inducing Signaling Complex (DISC): Ligand binding recruits adaptor proteins (e.g., FADD) and initiator caspases (e.g., caspase-8).
- Caspase Cascade: Caspase-8 activates effector caspases (e.g., caspase-3), which cleave cellular components, leading to cell death.
The discussed pathway cannot be avoided in immunological processes of removing cells, among which is the elimination of virus-containing cells.
Meet the Molecule: Caspase-8
Caspase-8 was discovered in the 1990s. It is the key initiator caspase in the extrinsic pathway. It acts as the first domino in the cascade that dismantles the cell.
2. The Intrinsic Pathway: Stress from Within
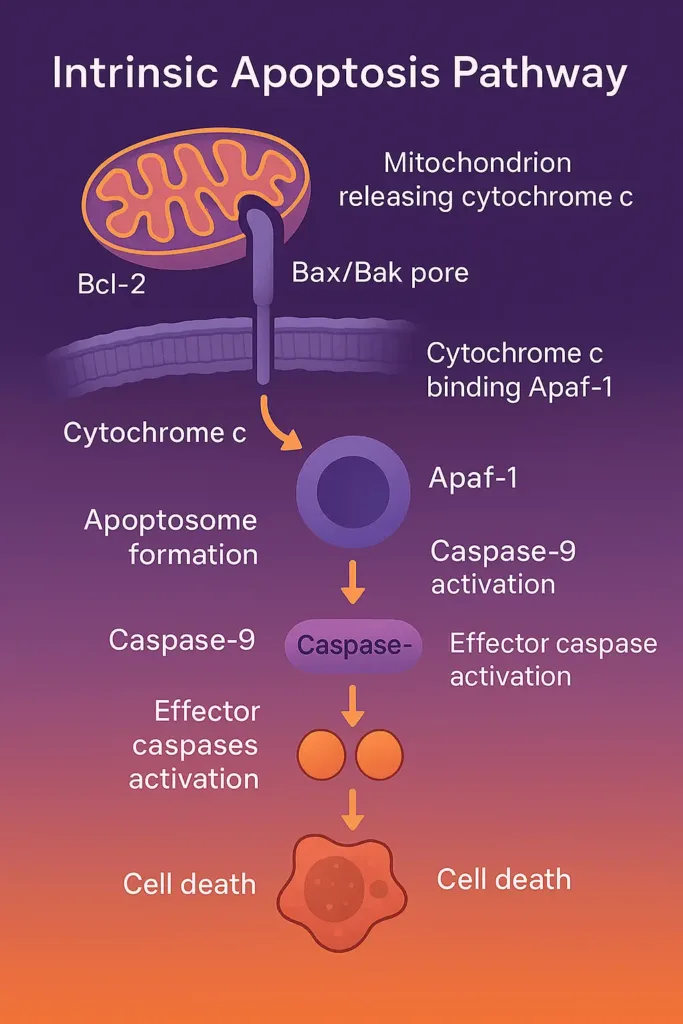
The intrinsic pathway is activated in response to internal or intrinsic stressors. These stressors include DNA damage or the removal of growth factors. In this process, the communication in the pathway is passed on by the mitochondria.
- Bcl-2 Family Proteins: These include pro-apoptotic (e.g., Bax, Bak) and anti-apoptotic (e.g., Bcl-2, Bcl-xL) members. Bax and Bak form pores in the mitochondrial outer membrane, releasing cytochrome c.
- Apoptosome Formation: Cytochrome c binds Apaf-1, forming the apoptosome, which activates caspase-9.
- Caspase Cascade: Caspase-9 activates effector caspases, leading to cell dismantling.
The elimination of cells by the mechanism of apoptosis is crucial. This process occurs before the cells enter into necrosis. It is important for protecting the organism against diseases like cancer.
Meet the Molecule: Bcl-2
Bcl-2 was initially discovered during the development of the B-cell lymphoma. It was hence christened the guardian of life due to its anti-apoptotic properties. These properties inhibited apoptosis by sequestering pro-apoptotic Bak and Bax. Surpassing amounts of Bcl-2, nonetheless, impose a pro-oncogenic phenotype.
3. The Executioners: Caspases and Their Targets
Caspases (cysteine-aspartic proteases) are the executioners of apoptosis. Synthesized as inactive zymogens, they are activated by cleavage and target:
- Nuclear Lamins: Causing nuclear breakdown.
- Cytoskeletal Proteins: Leading to cell shrinkage and blebbing.
- DNA Repair Enzymes: Promoting DNA fragmentation.
This cascade ensures the cell is irreversibly dismantled.
Apoptosis in Health and Disease
Programmed cell death — that’s a big deal for keeping our bodies functioning properly. It is like, without it, things would get quite messy. But here is the catch. Apoptosis is just a fancy term for this process. When it does not work as it should, it can lead to some highly serious health issues.
A. Cancer Prevention & Therapies
Apoptosis is used by healthy somatic cells to remove precancerous cells before a tumor occurs. In most interplays linked to cancer, the apoptotic cascade is inhibited. This inhibition raises questions about the potential success of resurrecting the self-directed death of the cell. The mechanism of action of current chemotherapeutic regimens includes the start of the intrinsic apoptotic pathway with DNA-damage agents. Similarly, new immunotherapies, especially CAR-T cell-based methods, trigger the extrinsic pathway of apoptosis, causing cancer cell-specific cell death.
B. Neurodegenerative Diseases
Too much apoptosis may have a negative impact on the brain. Take Alzheimer’s disease, for example. In that case, nerve cells are dying off excessively. This excess is a big part of why folks start losing their memories. And it doesn’t stop there. Other conditions like Parkinson’s and Huntington’s diseases also show this tendency toward excessive apoptosis in certain areas of the brain.
The interesting thing is that researchers are working hard to find ways to balance this out. Trying to tweak apoptosis so we can actually protect those precious neurons. It is a totally fascinating area of study, for sure!
C. Autoimmune Disorders
When the body does not get rid of those self-reactive immune cells, it can lead to autoimmunity. Take lupus or rheumatoid arthritis, for example. In these conditions, there is a glitch in the apoptosis process. This is the way our cells are supposed to die off when they are no longer needed. Because of this glitch, those rogue immune cells hang around and start attacking healthy tissues, which is obviously not good. But the good news? There are therapies in the study phase. They aim to fix this issue. The goal is to restore the normal process of cell death in the immune system. So, fingers crossed for some breakthroughs!
| Disease/Condition | Associated Apoptosis Pathway | Impact of Dysregulation | Therapeutic Strategy |
|---|---|---|---|
| Cancer (e.g. leukemia, melanoma) | Intrinsic and Extrinsic | Apoptosis is suppressed; damaged or abnormal cells survive and proliferate | Chemotherapy, radiation, immunotherapy (e.g. CAR-T cells) |
| Alzheimer’s Disease | Intrinsic | Excess apoptosis leads to progressive neuron loss | Neuroprotective agents, caspase inhibitors (under research) |
| Parkinson’s Disease | Intrinsic | Dopaminergic neurons die due to mitochondrial stress and apoptosis | Antioxidants, mitochondrial stabilizers |
| Systemic Lupus Erythematosus (SLE) | Extrinsic / Defective Clearance | Faulty apoptosis allows self-reactive immune cells to persist | Immunosuppressants, therapies restoring apoptosis signaling |
| HIV Infection | Extrinsic | HIV triggers apoptosis of CD4+ T-cells, weakening the immune system | Antiretrovirals, immune modulators |
Glossary of Terms
- Apoptosis: Programmed cell death characterized by controlled cellular dismantling without inflammation
- Caspases: Proteolytic enzymes that execute apoptosis by cleaving specific cellular proteins
- Apoptosome: A large protein complex that activates caspase-9 in the intrinsic pathway
- DISC (Death-Inducing Signaling Complex): A protein complex formed at death receptors that activates caspase-8
- Cytochrome c: A protein normally involved in cellular respiration that triggers apoptosis when released from mitochondria
- Bcl-2 proteins: A family of proteins that regulate apoptosis, including both pro-apoptotic and anti-apoptotic members
- Necrosis: Uncontrolled cell death resulting from injury or disease, characterized by inflammation
- Necroptosis: A regulated form of cell death that shares features with both apoptosis and necrosis
- Fas ligand: A death signal that binds to Fas receptors to trigger the extrinsic pathway
- Mitochondrial outer membrane permeabilization (MOMP): A critical event in the intrinsic pathway that releases apoptotic factors.
Frequently Asked Questions
Apoptosis is well-categorized and orderly. There is no inflammation after it. In the opposite case, necrosis occurs abruptly and randomly and tends to trigger an inflammation.
Binding of outside signals to death receptors forms DISC complex. The DISC complex turns on initiator caspases, which trigger the cascade of cell-death.
Apoptosome is initiated by the leakage of cytochrome c out of mitochondria. This is a significant procedural process of the intrinsic apoptosis process.
Apoptosis stops when the stress signals diminish in early apoptosis. However, cell death is irreversible after caspases overcome a certain threshold.
The Future of Cellular Life and Death
Programmed cell death is very important—it is like this carefully controlled process our bodies have going on. Cells can actually choose to self-destruct through two main procedures: intrinsic and extrinsic apoptosis. It is quite fascinating how they do this in such an organized manner, using energy to make it all happen. This whole process is essential for keeping our tissues in tip-top shape. It shapes our organs just right. It also protects us from diseases.
But if apoptosis does not work the way it should, it can lead to some serious problems. We are talking about life-threatening cancers or degenerative disorders that can really mess things up. So, yeah, it is crucial for our health!
References
- NCBI Bookshelf, “Apoptosis – StatPearls,” 2025.
- Nature, “The BCL2 Family: From Apoptosis Mechanisms to New Advances,” 2025.
- Taylor & Francis Online, “A Comprehensive Insight into Apoptosis,” 2025.
- Nature Reviews Molecular Cell Biology – Comprehensive review articles.
- Annual Review of Cell and Developmental Biology – Yearly updates on apoptosis research.