Cells are the basic building blocks of our body. When these small cells come together, our body is formed. Cells work like a small factory inside our body, where each cell has its own job and performs its job well. When we are healthy, our cells also work easily, which means that the working of the cells of a healthy person is also smooth. If a person falls ill, diseases affect the cell structure and functioning, so everything changes. Due to which the cells either become weak or stop doing their work completely, due to which various diseases arise in the person.
Here it is very important to understand how diseases affect the cell structure, which disrupts the functions of cells, because this is the main reason. When we understand the basics, then it will be easy for us to treat any disease because these microscopic meanings are also the reasons due to which major diseases such as cancer arise. This knowledge can help us develop the best treatments for diseases and the best strategies to control such diseases.
How Diseases Disrupt Cell Structure
Diseases can wreak havoc on cell structure in several ways. For example, viruses hijack the cell’s machinery to reproduce, harm, or destroy the cell in the process. On the other hand, bacterial infections can produce toxins that weaken cell membranes or disrupt organelle operation. On the other hand, cancer causes cells to grow uncontrollably, leading to abnormal cell structures and impaired performance.
The Cytoskeleton Under Attack
The cytoskeleton acts like an internal scaffolding within the cell. This network of protein fibers gives cells their shape and helps move materials around. When a disease attacks the cytoskeleton, cells lose their structure and function.
1. F-Actin Dysregulation and Cell Shape
Actin filaments act like flexible cables throughout the cell. They contract and expand to help cells move and maintain their proper shape. These protein strands are especially important in muscle cells, blood cells, and the cells that connect our organs.
But when there is a malfunction within the actin filaments, several serious conditions can arise. In some blood disorders, red blood cells become misshapen and can’t carry oxygen effectively. This happens because the actin network that normally keeps these cells flexible breaks down into a disk-like shape.
Cataracts provide another clear example of problems with F-actin. The lens of your eye contains cells filled with actin filaments. When these filaments clump together or break, the lens becomes cloudy. This process often occurs gradually with age, which is why cataracts are common in older adults.
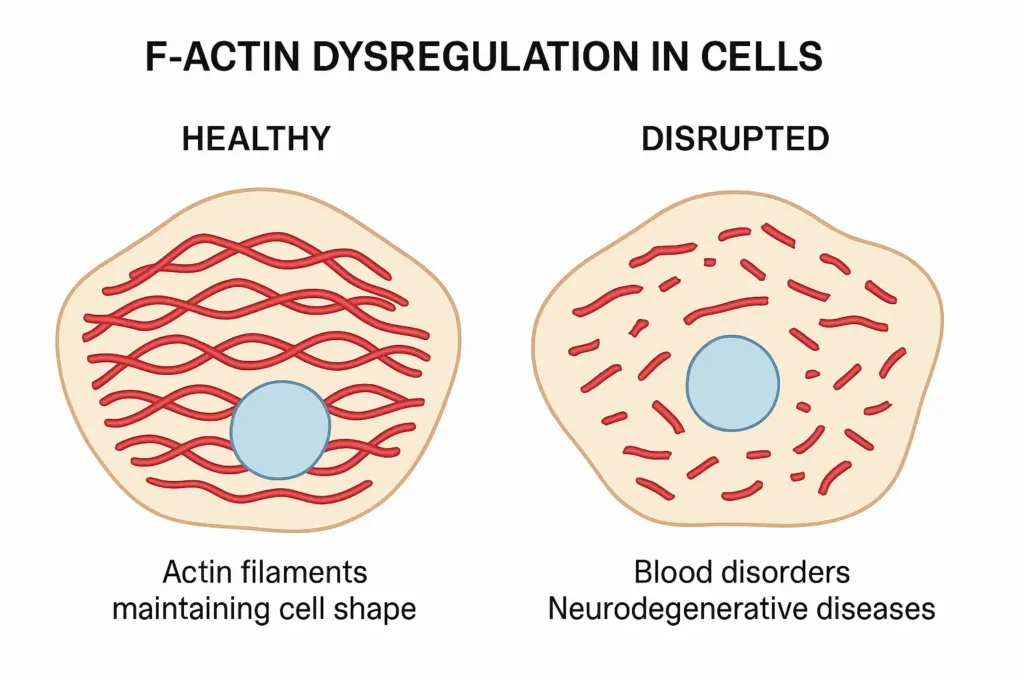
Osteoarthritis and tendinosis also involve actin dysregulation. In healthy cartilage and tendons, cells maintain tissue structure through properly organized actin networks. When disease disrupts these networks, cells are unable to effectively repair tissue damage, resulting in joint pain, stiffness, and decreased mobility.
2. Microtubules and Intermediate Filaments in Disease
Let me explain to you in simple terms that microtubules act like highways within cells, transporting materials from one place to another. Intermediate filaments provide structural support, especially in cells that experience mechanical stress. Together, these components maintain cellular organization and enable proper function.
Degenerative diseases are the clearest examples of how microtubule disruption affects health. In Alzheimer’s disease, a protein called tau normally helps stabilize microtubules in brain cells. When tau becomes abnormal, the microtubules fall apart. Brain cells can’t move nutrients and waste products efficiently, leading to cell death and memory loss.
Parkinson’s disease involves similar microtubule problems, but primarily affects the brain cells that produce dopamine. As these cells lose their microtubule network, patients experience tremors, slow movement, and balance problems. The progressive nature of these diseases reflects the gradual breakdown of cellular transport systems.
For readers seeking an in-depth understanding of cellular structure, “Molecular Biology of the Cell” provides comprehensive coverage of cytoskeletal components and their roles in health and disease.
Viruses: The Cellular Hijackers

When a virus infects a cell, it controls the cell’s machinery to replicate itself. This process often leads to significant structural changes. Cell membranes can become compromised, allowing more viruses to enter and exit the cell. Inside the cell, organelles are typically reprogrammed to aid in virus production, causing their malfunction or destruction. These changes can result in cell death and contribute to the spread of the virus within the body.
The influenza virus, for example, attaches to lung cells. It then injects its genetic material inside. The normal functions of the cell stop as it becomes the virus causing factory. Eventually, the cell bursts, releasing new viruses to infect neighboring cells.
Viral Cytopathic Effects (CPE) on Cells
When viruses invade cells, they don’t just hide quietly. They cause visible changes called viral cytopathic effects. These changes help doctors identify viral infections and understand how viruses damage tissues.
1. Morphological Changes in Infected Cells
Under a microscope, virus-infected cells look dramatically different from healthy cells. One of the most common changes is cell rounding. Normally, cells have specific shapes that are suited to their functions. Infected cells often become round and eventually separate from the surrounding tissue.
Syncytia formation shows another striking change. Some viruses cause individual cells to fuse together, forming larger multicellular cells. This process is particularly prominent in respiratory infections, where lung cells fuse and lose their barrier function.
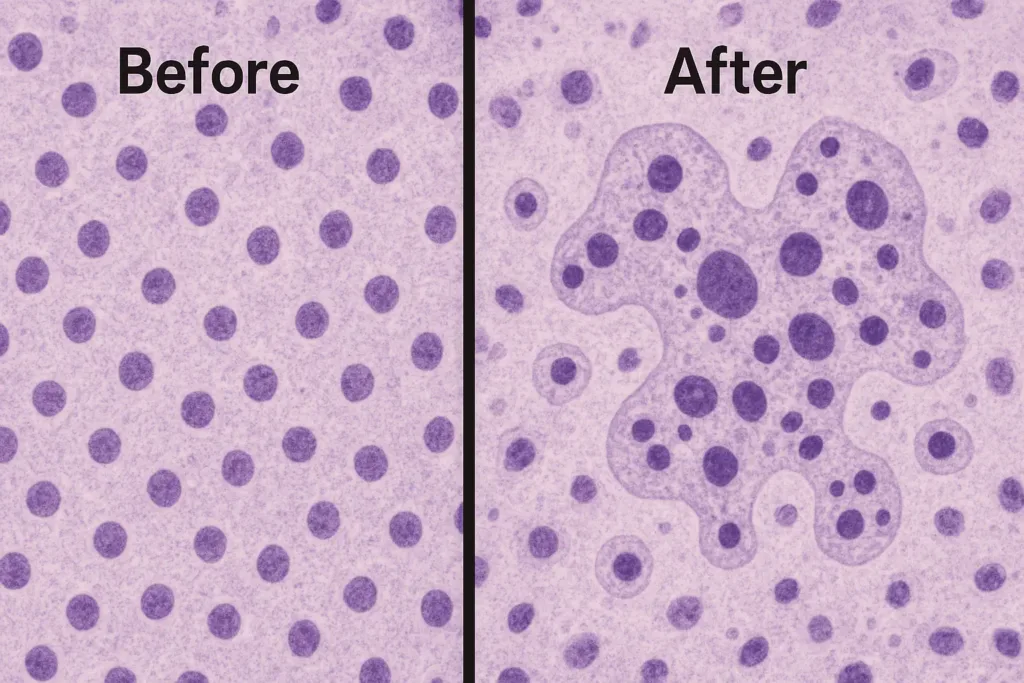
Inclusion bodies appear as dark spots within infected cells. These structures contain viral components and represent areas where viruses replicate. Different viruses produce characteristic inclusion patterns that help pathologists identify specific infections.
The microscopy studies referenced in the table below illustrate how different families of viruses produce distinct cytopathic patterns, ranging from cell shrinkage to membrane blebbing.
| Virus Family | Primary CPE Pattern | Cell Morphology Changes | Timeline | Common Examples |
|---|---|---|---|---|
| Adenovirus | Inclusion bodies | Cell rounding, nuclear enlargement | 24-48 hours | Common cold, conjunctivitis |
| Herpesvirus | Syncytia formation | Cell fusion, multinucleated giants | 12-24 hours | HSV-1, HSV-2, CMV |
| Influenza | Cell shrinkage | Membrane blebbing, apoptosis | 18-36 hours | Seasonal flu, H1N1 |
| Coronavirus | Membrane damage | Cell swelling, organelle disruption | 24-72 hours | COVID-19, SARS, MERS |
| Poliovirus | Cytoplasm clearing | Cell lysis, rapid breakdown | 6-12 hours | Poliomyelitis |
| Paramyxovirus | Syncytia clusters | Large multinucleated cells | 24-48 hours | Measles, mumps, RSV |
2. Physiological and Biochemical Alterations
In addition to the visible changes, there are many other viral cytopathic effects that disrupt basic cellular processes. Protein synthesis is often shut down because viruses redirect the cellular machinery to produce viral components instead of normal proteins. This change tells us why infected individuals feel weak and tired.
Metabolism also changes dramatically during viral infections. Cells normally burn glucose efficiently to produce energy. Infected cells often switch to less efficient methods of energy production, leading to fever and fatigue. Some viruses actually manipulate cellular metabolism to promote viral reproduction, which can damage cellular components through oxidative stress.
Bacterial Toxins and Cell Damage
Bacteria can produce toxins that directly target cell structures. For example, certain toxins can pierce the cell membrane, causing the cell to lose its integrity and die. Other toxins can disrupt the operation of the cell’s energy producers. They affect components like mitochondria. This leads to decreased energy production and cell death. These structural damages can impair tissue operation and cause symptoms of bacterial infection.
Organelle Dysfunction in Disease
Organelles are specialized structures within cells that perform specific functions. When organelle dysfunction occurs, these cellular components are unable to do their job properly, leading to a wide range of health problems.
1. Golgi Apparatus and Protein Trafficking Disorders
The Golgi apparatus acts like a cellular post office, modifying and packaging proteins for delivery throughout the body. This organelle consists of stacked membranes that process proteins in an assembly-line fashion.
Golgi fragmentation occurs in several diseases, including some neurological conditions and cancer. When the Golgi is damaged, cells can’t properly process or transport proteins. This causes proteins to accumulate inside cells and reduces the delivery of proteins to tissues that need them most.
Some genetic disorders directly affect Golgi function. These conditions often involve problems with protein glycosylation – the process of adding sugar molecules to proteins. Without proper glycosylation, proteins can’t fold properly or reach their intended destination. For more details check out our article: Golgi Apparatus and Protein Synthesis.
2. Mitochondrial Dysfunction and Energy Crisis
Mitochondria are the power plants of the cell, producing the energy currency, ATP. Mitochondrial dysfunction leads to an energy crisis that can affect any tissue but is particularly devastating for high-energy organs such as the brain, heart, and muscles. Mitochondrial diseases, often genetic, can cause a wide range of symptoms, from muscle weakness to neurological deficits. Furthermore, compromised mitochondrial function is a key factor in many metabolic syndromes, including type 2 diabetes and obesity.
To understand the complex pathways of cellular energy production and their link to disease, a resource such as Introduction to Genetics: A Molecular Approach provides an authoritative and detailed guide.
3. Lysosomal Storage Diseases
Lysosomes are the recycling centers of the cell, using enzymes to break down waste materials. In lysosomal storage diseases, a genetic defect blocks the production of a specific enzyme. As a result, undigested molecules accumulate inside the lysosomes, causing them to swell and disrupt cell function. This buildup can damage organs and tissues, causing a wide range of symptoms. For example, in Tay-Sachs disease, a fatty substance builds up in the nerve cells of the brain, leading to nerve damage.
Cancer and Abnormal Cell Growth

Cancer signifies a unique threat to cell structure. It occurs when normal cells mutate and grow uncontrollably. These rogue cells ignore the normal growth signals of the body and avoid programmed cell death.
Cancer cells often show abnormal shapes and sizes. Their internal structures become disorganized. As tumors grow, they can compress and damage surrounding healthy cells, disrupting normal tissue operation.
Autoimmune diseases and cellular attack
Autoimmune diseases occur when the body’s immune system mistakenly attacks its own cells. This attack can lead to various structural changes. For example, in rheumatoid arthritis, the immune system targets cells in the joints. This targeting causes inflammation and damage to cell membranes. It also impacts the underlying structures. This leads to pain, swelling, and a decrease in joint operation. These structural changes in cells can significantly lower the quality of life for individuals. People suffering from autoimmune diseases face these challenges.
Chromatin and Nuclear Reorganization
The cell nucleus contains genetic material that is organized into chromatin—a complex of DNA and proteins. Disease can dramatically alter the structure of chromatin, affecting how genes are expressed and regulated.
Genome Architecture Changes
The 3D arrangement of chromatin within the nucleus is not random. It plays an important role in gene regulation. In conditions such as tendinosis, excessive mechanical stress on tendon cells can cause massive chromatin reorganization. This process, which often involves histone modifications and repatterning of DNA loops, can alter gene expression, leading to the production of proteins that promote inflammation and tissue degradation rather than repair.
Epigenetic Alterations: Beyond DNA sequencing, chemical tags can be added to DNA or histones, which alter how genes are read. These epigenetic changes are increasingly recognized for their role in disease. For example, in some forms of cancer, epigenetic changes can silence tumor suppressor genes or activate oncogenes, promoting uncontrolled cell growth.
Neuron degenerative Diseases and Cellular Degeneration
Neuron degenerative diseases, like Alzheimer’s and Parkinson’s involve the gradual loss of structure and role in neurons. In Alzheimer’s disease, abnormal protein deposits form plaques and tangles within neurons, disrupting their communication and leading to cell death. In Parkinson’s disease, the loss of dopamine producing cells impacts motor control. Structural changes in these diseases are often progressive, leading to a decline in cognitive and motor operation over time.
Real-World Disease Examples
To put these cellular mechanisms into a broader context, let’s look at some real-world examples.
Blood Disorders and Cytoskeletal Failure
As discussed, hereditary spherocytosis illustrates a direct link between a defective cytoskeleton and debilitating disease. The genetic mutation leads to defective spectrin protein, which weakens the interior of the red blood cell and causes it to lose its elasticity. This simple structural change has a cascade of effects, from anemia to possible spleen damage, highlighting how a microscopic defect can manifest as a systemic disease.
Viral Infections and Cytopathic Effects
The common cold, caused by rhinovirus, is a classic example of CPE. The virus causes respiratory epithelial cells to swell and divide, leading to symptoms of a stuffy nose and sore throat. Although often mildly uncomfortable, it is a powerful example of a virus directly altering cell morphology to achieve its replication goals.
Neurodegenerative Disorders and Organelle Dysfunction
In Alzheimer’s, brain cells are starving. The breakdown of the microtubule-based transport system, caused by tau protein tangles, means that mitochondria and other essential nutrients can’t reach the periphery of neurons. This is a clear case of organelle (mitochondria) dysfunction resulting from cytoskeletal (microtubule) failure, leading to a massive energy crisis and ultimately the death of the cells that power memory and thought.
Conclusion
From the structural integrity of the cytoskeleton to the energy production of mitochondria and the intricate organization of the cell genome, diseases have a profound impact on every level of cell structure and function. We have seen how a single mutation or microscopic invader can set off a chain reaction that disrupts the cell’s architecture, its internal transport system, and its genetic programming.
Understanding these changes at the cellular level is more than just an academic exercise. It is crucial for unlocking new therapeutic strategies. By targeting specific molecular pathways that cause cytoskeleton breakdown, organelle failure, or chromatin reorganization, scientists and doctors can develop more effective treatments that address not just the symptoms but the underlying cause of the disease. This area of research holds great promise for the future of medicine.
Recommended Reads for Curious Minds
- Molecular Biology of the Cell (7th Edition) by Bruce Alberts
- BIOLOGY Super Easy by Marcus L. Trevors
- The Immortal Life of Henrietta Lacks by Rebecca Skloot
Frequently Asked Questions
Early cellular changes often appear before symptoms become noticeable. These include altered cell shape, changes in organelle appearance under microscopy, and disrupted protein production. In many cases, blood tests can detect cellular dysfunction markers like elevated enzymes or abnormal protein levels before patients experience obvious symptoms.
Yes, many cells have remarkable repair abilities, but this depends on the type and extent of damage. Cells can replace damaged organelles, rebuild cytoskeletal networks, and restore normal function if the underlying cause is addressed. However, severe damage to critical structures like DNA or extensive mitochondrial dysfunction may be irreversible.
Disease specificity often relates to cellular characteristics and requirements. For example, neurons are particularly vulnerable to mitochondrial dysfunction because they have high energy demands. Similarly, rapidly dividing cells are more susceptible to DNA damage, while cells with extensive cytoskeletal networks may be more prone to structural disorders.